Endless Improvements
العربية
Mineral Base Oil Manufacturing
Crude oil in its natural state has no value to consumers and must be transformed into products that can be used in the marketplace. Various physical and chemical methods are used in refining processes. Heat, pressure, catalysts, and chemicals are applied under widely varying process designs, operating conditions, and chemical reactions to convert crude oil and other hydrocarbons into petroleum products.
1. Distillation
a. Atmospheric Distillation
Distillation is the process of separating the component substances of a liquid mixture of two or more chemically discrete substances. The separation process is realized by way of selective boiling of the mixture, then condensation of the vapors in a still.
1.
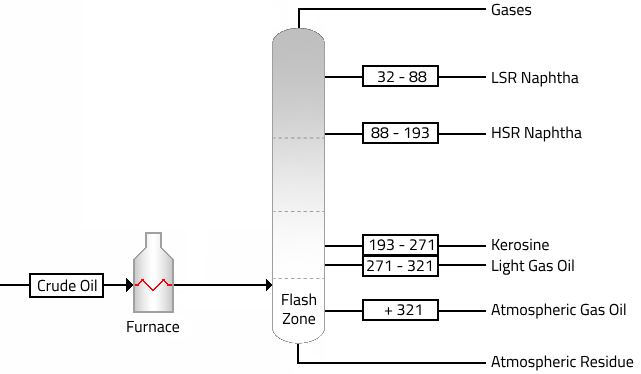
The crude oil is heated to about 399°C in a furnace and charged to the flash zone of the atmospheric distillation unit. The furnace discharge temperature is sufficiently high to cause vaporization of all products withdrawn above the flash zone.
2.
Inside the column, a variety of different mechanical arrangements are used to assist separation of different boiling range fractions:
Trays: to allow rising vapor and falling liquid to contact each other and come to equilibrium.
Packing: giving excellent contact and distribution of liquid and vapor.
The main products from a typical crude distillation unit are as follows:
| Fractions | Description | B.P. °C |
| Gases | The stream contains propane and butanes as well as methane and ethane. | |
| Light straight-run Naphtha | The stabilized LSR naphtha stream is desulfurized and used in gasoline blending. | 32 – 88 |
| Heavy straight-run Naphtha | Used as catalytic reformer feed to produce high-octane reformate for gasoline blending and aromatics. | 88 – 193 |
| Kerosine | It is widely used as a fuel in aviation as well as households. | 193 – 271 |
| Light Gas Oil | The light and atmospheric oils are processed in a hydrocracker or catalytic cracker to produce gasoline, jet, and diesel fuels. | 271 – 321 |
| Atmospheric Gas Oil | + 321 |
b. Vacuum Distillation
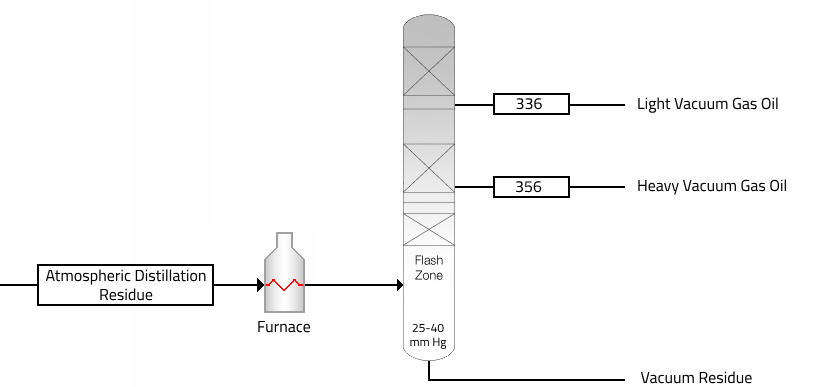
The temperatures required for atmospheric distillation of the heavier fractions of crude oil are so high that thermal cracking would occur. These materials are, therefore, distilled under vacuum, because the boiling temperature decreases with a lowering of the pressure. Distillation is carried out with absolute pressures in the tower flash zone area of 25 to 40 mmHg.
The main products from a vacuum crude distillation unit are as follows:
| Fractions | Product | Withdraw Temp. | EPB °C |
| Light Vacuum Gas Oil | Produce high-octane reformate for gasoline blending and aromatics. | 336 | 462 |
| Heavy Vacuum Gas Oil | Feedstocks for lubricating oil processing units. | 356 | 542 |
| Vacuum Residue | Produce heavy and Lube base stock. | + 356 | + 542 |
2. De-asphalting
Light and Heavy vacuum Gas Oil for producing lubricating oil base stocks can be sent directly to the rest of manufacturing processes, but Vacuum Residue requires de-asphalting to remove the asphaltenes and resins before undergoing the rest of manufacturing processes.
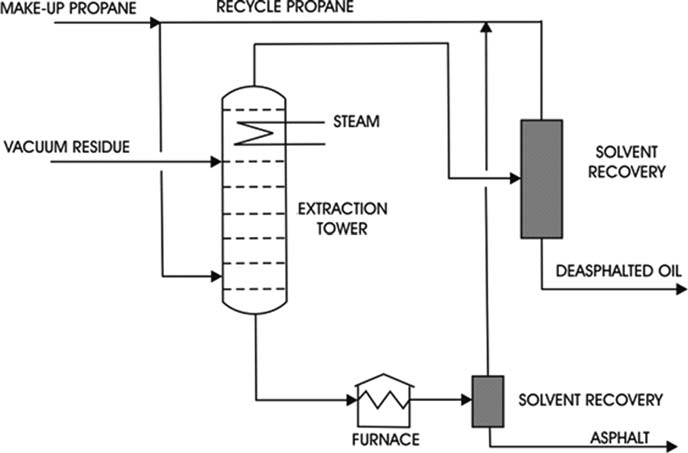
Propane De-asphalting:
Propane has unusual solvent properties in that from 40 to 60°C paraffins are very soluble in propane, but the solubility decreases with an increase in temperature until at the critical temperature of propane 96.8°C all hydrocarbons become insoluble. In the range of 40 to 96.8°C, the high molecular-weight asphaltenes and resins are largely insoluble in propane.
The feedstock is contacted with four to eight volumes of liquid propane at the desired operating temperature. The heavier the feedstock, the higher the ratio of propane to oil required.
1.
Propane injects into the bottom of the extraction tower, and the Vacuum Residue enters near the top of the tower. As the propane rises through the tower, it dissolves the oil from the residuum and carries it out of the top of the tower.
2.
The asphaltene and resin phase leaving the bottom of the tower is the raffinate, and the propane–oil mixture leaving the top is the extract.
3.
The solvent recovery system of the propane de-asphalting process is used to recover the propane from the raffinate and extract phases.
The asphalt recovered from the raffinate can be blended with other asphalts into heavy fuels or used as a feed to the coking unit.
The heavy oil product from extract is called bright stock. It is a high viscosity blending stock that, after further processing, is used in the formulation of heavy-duty lubricants for truck, automobile, and aircraft services.
3. Improving the Viscosity Index
Removing of the aromatic compounds from the lubricant oil base stock leads to improve the viscosity index and the oxidation resistance of the lubricants.
| Hydrocarbon | VI |
| n-Paraffins | 175 |
| i-Paraffins | 155 |
| Mononaphthenes | 142 |
| Dinaphthenes+ | 70 |
| Aromatics | 50 |
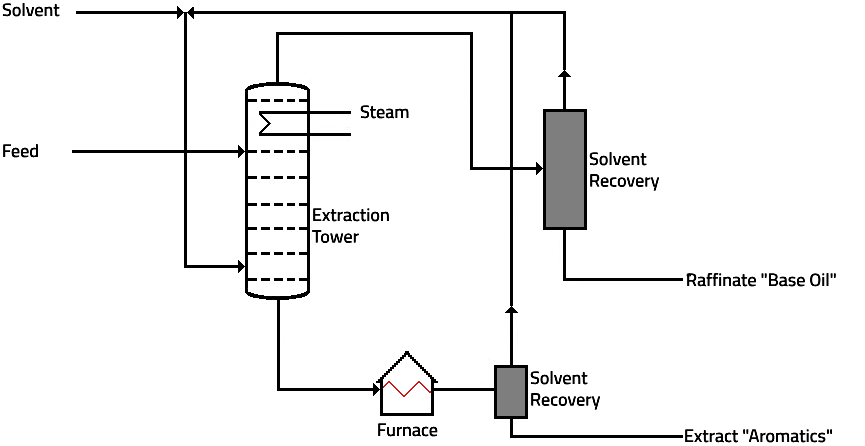
a. Solvent Extraction
The solvent selectively dissolves the undesired aromatic components, the extract, leaving the desirable saturated components, especially alkanes, as a separate phase, the raffinate. There are three solvents used for the extraction of aromatics from lube oil feedstocks furfural, phenol, and N-methyl-2-pyrrolidone (NMP).
The process flow through the furfural extraction unit is similar to that of the propane de-asphalting unit.
1.
The light, heavy vacuum gas oil, or de-asphalted oil feed is introduced near the bottom of the extraction tower and the solvent enters near the top.
2.
The oil-rich phase rises through the tower, and the solvent-rich phase descends the tower. Trays or packing are used to provide intimate contact between the two phases.
3.
At the end of the extraction process the result is two product streams:
The raffinate stream: is mainly extracted oil containing a limited amount of solvent.
The extract stream is: a mixture of solvent and aromatic components.
b. Hydrocracking
Components of lubricating oil fractions that have high viscosity indices are the mononaphthalenes and isoparaffins. Hydrocracking of vacuum gas oils increases the paraffin concentration and the viscosity index of a lube oil feedstock and produces increasing quantities of the mononaphthalenes and isoparaffins as hydrocracking severity increases. Such a base oil has advantages over ordinary hydrotreated or extracted base oils for its viscosity index is higher and volatility is lower than that for a conventionally produced oil of the same viscosity. These are important benefits for formulating automotive engine lubricants.
Hydrocracking reaction mechanism consists of:
Hydrogenation: addition of hydrogen to carbon–carbon double bond.
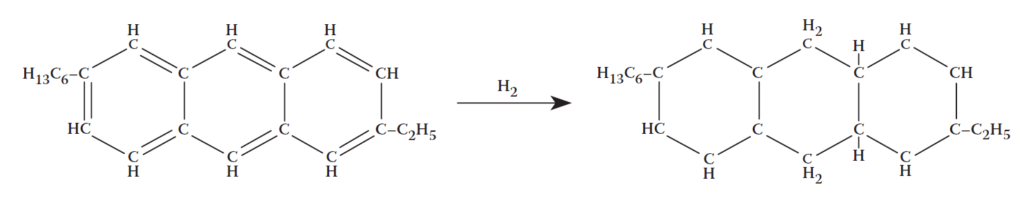
Catalytic cracking: scission of a carbon–carbon single bond.
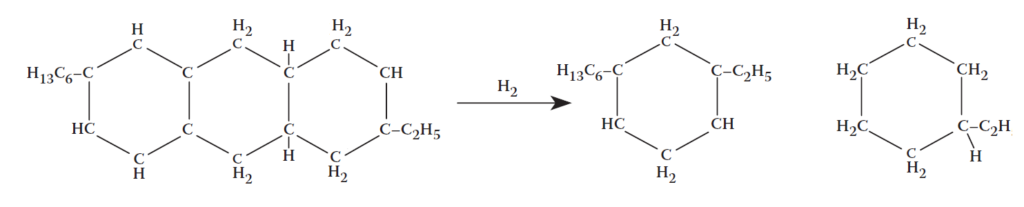
Further processing of the hydrocracker residue is needed . Extraction is desirable to remove traces of polycyclic aromatics and improve product quality.
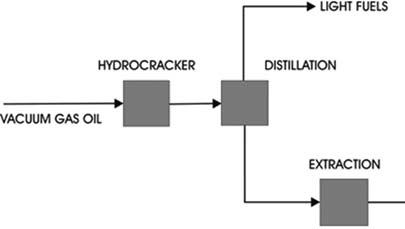
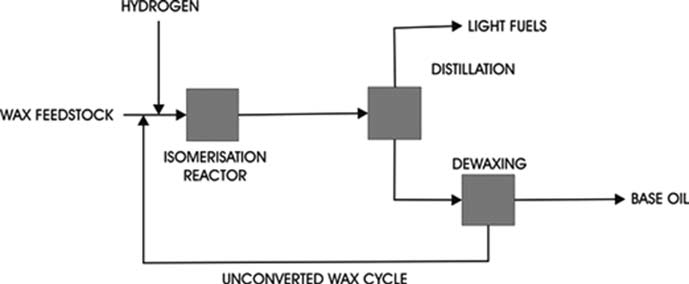
4. De-waxing
The material which crystallizes out of solution from lubricant distillates or raffinates is known as wax. Wax content is a function of temperature; as the temperature is reduced, more wax appears. Sufficient wax must be removed from each base oil fraction to give the required low-temperature properties for each base oil grade.
| Hydrocarbon | Crystallization Behavior |
| n-Paraffins | Easily crystallize in the form of large crystals [predominant in the lighter distillates]. |
| i-Paraffins | Forms smaller crystals [predominate in the heavier fractions]. |
| Naphthenes | Relatively free of wax. |
There are two types of processes in use today. One uses refrigeration to crystallize the wax and solvent to dilute the oil portion sufficiently to permit rapid filtration to separate the wax from the oil. The other uses a selective hydrocracking process to crack the wax molecules to light hydrocarbons.
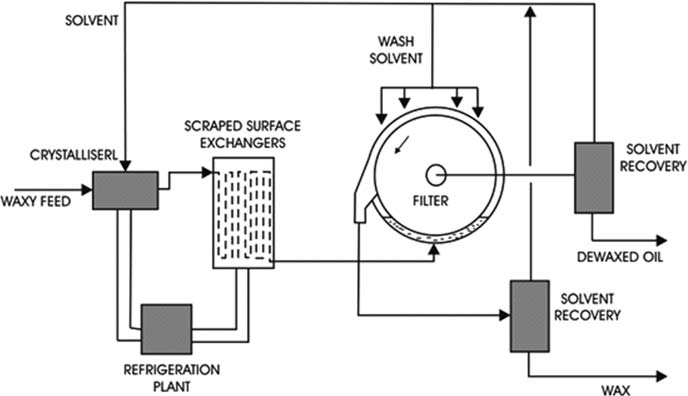
a. Solvent De-waxing
1.
Solvent and oil are mixed together.
2.
The mixture progressively chilled to the required temperature for filtration, which will be several degrees lower than the desired pour point. Chilling takes place in special heat exchangers with mechanically driven scrapers to keep the pipe walls clear of solidifying wax, aid heat transfer and ensure that the oil/wax/solvent slurry remains mobile.
3.
Filtration is carried out on large rotary drum filters with suction applied to the inside of the horizontally mounted drum which slowly rotates with the lower part of the drum immersed in the chilled slurry. As oil passes through the filter cloth, a layer of wax (about 0.5 cm thick) builds up on the cloth and is later removed by a scraper blade or blown off by inert gas as the drum rotates.
4.
The de-waxed oil/solvent and the crude wax are handled as separate streams for solvent recovery.
5.
The wax contains an appreciable amount of oil because filtration pressures are very low and this oil can be removed by a second filtration stage which also yields high-quality treatment wax products.
b. Catalytic De-waxing
A special shape-selective catalysts are used to selectively hydrocrack only the straight-chain alkanes to low boiling point by-products. De-waxing catalysts are based on molecular sieves such as zeolites or silicalite, which have highly porous structures based on a regular arrangement of channels. The channel openings have diameters of 50–70 nm which will admit only linear or very lightly branched alkanes. Non-waxy branched alkanes and cyclic structures cannot enter the pore structure and so are not converted, although long alkyl side chains on aromatic or alicyclic nuclei can be stripped off.
Catalytic de-waxing is highly selective for linear alkanes and, since these molecules have the highest VI characteristics, it is usual for catalytically de-waxed base oil to have lower VIs than solvent de-waxed oils of the same pour point.
c. Iso de-waxing
1.
The wax feedstock reacts over a catalyst in a hydrogen atmosphere but despite control of conditions to favor isomerization, a significant amount of cracking to lighter products is inevitable.
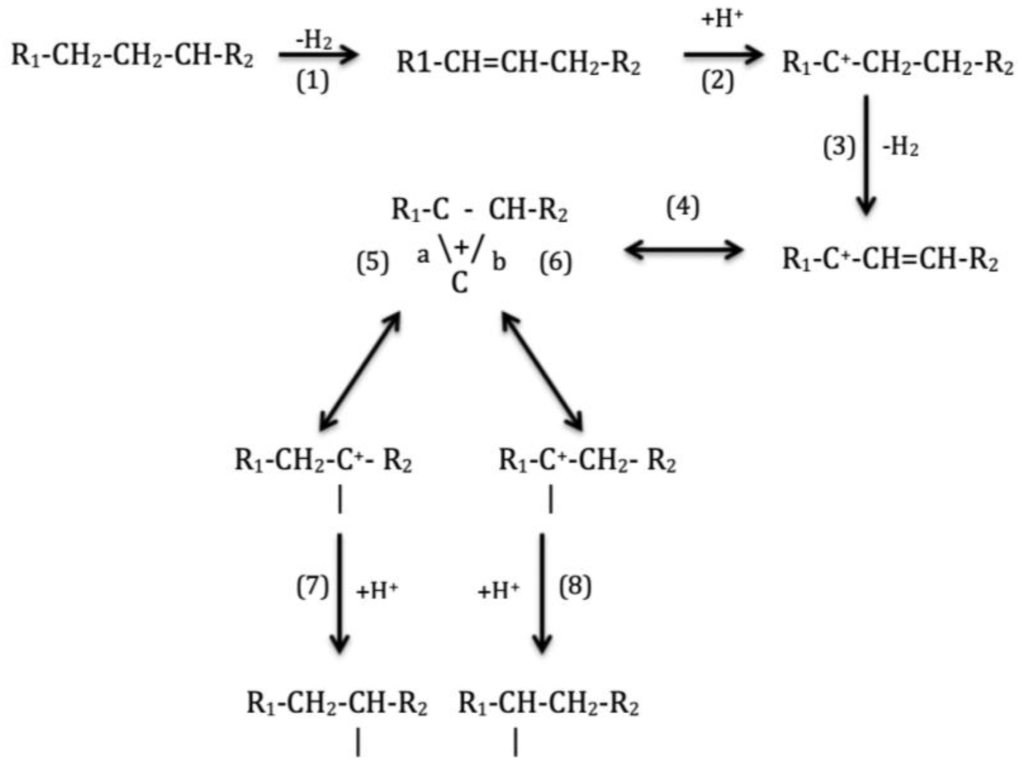
2.
Products are separated by distillation.
3.
The lubricant boiling range material is conventionally de-waxed.
Whereas the conventional catalytic de-waxing processes selectively hydrocrack wax to low molecular weight by-products, in iso-de-waxing the wax molecules are isomerized and remain within the boiling range of the base oil. The result is to improve both de-waxed oil yields and viscosity index by the retention of very high-VI iso-alkane molecules.
5. Finishing
Despite the intensive series of process steps carried out so far, trace impurities may still be present in the base oil. These undesirable components tend to be nitrogen-, oxygen- or, to a lesser extent, sulphur-containing molecules. A finishing step is needed to correct problems such as:
Poor color.
Poor oxidation or thermal stability.
Poor demulsification properties.
Poor electrical insulating properties.
a. Finishing by Clay Contacting
The contacting of the dewaxed oil with activated clays at elevated temperatures used to improve the stability of the finished oils in engine service. Polar compounds (aromatic and sulfur and nitrogen containing molecules) are adsorbed on the clay and removed by filtration. Spent clay disposal and operating restrictions have generally caused the clay treating to be replaced by hydrofinishing.
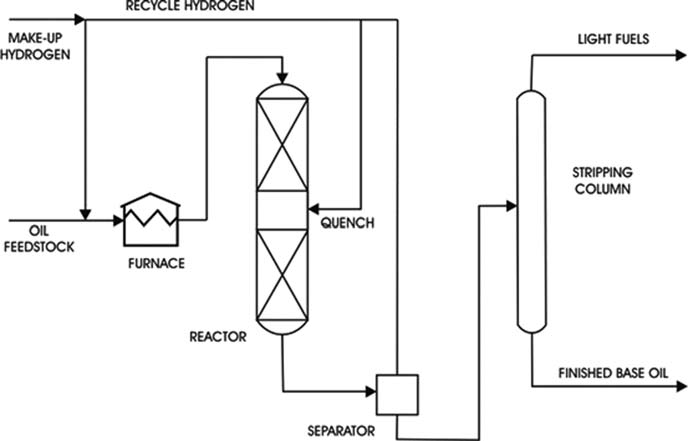
b. Hydro-finishing
Hydro-finishing depends on the selective, catalyzed hydrogenation of the impurities to form harmless products under relatively mild conditions. Hydro-finishing should be effective for removing organonitrogen molecules because they are largely responsible for poor color and stability of base oils, while organo-sulphur molecules should be retained because they tend to impart natural oxidation stability to the base oil.
1.
The hydrogen are pre-heated and the oil allowed to trickle downwards through a reactor filled with catalyst particles where hydrogenation reactions take place.
Typical reactor operating conditions for hydro-finishing are the following:
Catalyst temperature, 250–350°C.
Operating pressure, 20–60 bar.
Catalyst type, Ni, Mo supported on high-surface-area alumina particles.
2.
The oil product is separated from the gaseous phase and then stripped to remove traces of dissolved gases or water.

Facebook Comments